- Mon - Fri
- 9:00 am - 05:00 pm EST
- Closed on Weekends

XRF is an elemental analysis technique with unique capabilities including (1) highly accurate determinations for major elements and (2) a broad elemental survey of the sample composition without standards. For example, XRF is used in analysis of rocks and metals with an accuracy of ~0.1% of the major elements.
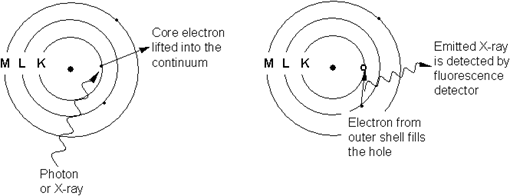
X-ray fluorescence (XRF) is the phenomenon where a material is exposed to X-rays of high energy, and as the X-ray (or photon) strikes an atom (or a molecule) in the sample, energy is absorbed by the atom. If the energy is high enough, a core electron is ejected out of its atomic orbital.
An electron from an outer shell then drops into the unoccupied orbital, to fill the hole left behind. This transition gives off an X-ray of fixed, characteristic energy that can be detected by a fluorescence detector. The energy needed to eject a core electron is characteristic of each element, and so is the energy emitted by the transition. The transition of an L shell electron dropping into the K shell is termed a Kα transition, while an M shell electron dropping into the K shell is a Kß transition.
When the energy source is a synchrotron, or the X-ray are focused by an optic, like a polycapillary, the X-ray beam can be very small and very intense, and atomic information on the sub-micrometer scale can be obtained.
Typically the lightest element that can be analyzed is beryllium (Z = 4), but due to instrumental limitations and low x-ray yields for the light elements, it is often difficult to quantify elements lighter than sodium (Z = 11).
1.Wavelength dispersive spectrometers (WDX or WDS): the photons are separated by diffraction on a single crystal before being detected.
2.Energy dispersive spectrometers (EDX or EDS): the detector allows the determination of the energy of the photon when it is detected.

Family owned and operated since 1979 (formerly known as EAC), we strive to give each customer the best service possible.
From Aerospace to Pharmaceuticals, we will work with you to tailor your specific analytical method.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.