- Mon - Fri
- 9:00 am - 05:00 pm EST
- Closed on Weekends

Neutron Activation Analysis (NAA) is one of the most sensitive analytical techniques used for multi-element analysis available today. The NAA procedure is capable of providing both quantitative and qualitative results for individual elements, with sensitivities that can be superior to those possible by any other analytical technique. Elemental Analysis Incorporated (EAI), as an innovator in the development and application of radio-nuclear chemistry analytical techniques, now offers its clients the ability to analyze some 75 individual elements (including certain organic elements) by NAA at trace and ultra-trace concentrations. Moreover, by developing scientific liaisons with selected nuclear reactor sites in North America, EAI is able to offer customers the expertise and capabilities of the premier scientists and research facilities available today. Combined with EAI’s tradition of excellence in customer service and technical assistance, EAI is uniquely positioned to assist clients with timely, cost-effective, and reliable trace element analysis for almost every conceivable field of industry or scientific research.
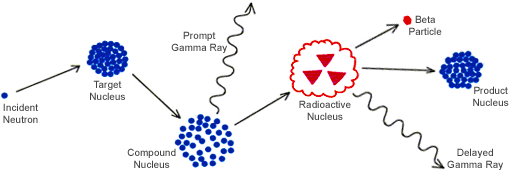
NAA is a physical technique that is based on nuclear reactions whereby the elemental content is determined by irradiating the subject sample with neutrons, creating radioactive forms of the desired element in the sample. As the sample becomes radioactive from the interaction of the neutron particle source and the nuclei of the element’s atoms, radioisotopes are formed that subsequently decay, emitting gamma rays unique in half-life and energy. These distinct energysignatures provide positive identification of the targeted element(s) present in the sample, while quantification is achieved by measuring the intensity of the emitted gamma rays that are directly proportionate to the concentration of the respective element(s) in the sample.
When neutrons activate the nucleus of the atom, it allows the total elemental content to be observed regardless of the oxidation state, physical location, or chemical form of the desired element. Since neutrons possess the ability to pass through most materials with little difficulty, this allows the center of the sample to become as radioactive as the surface, thereby reducing or even eliminating the potential for matrix effects. Because neutron activation can be applied to any element with an appropriate isotope, nearly 70% of elements in the Periodic Table can be analyzed by NAA.
Advantages of NAA: Unlike traditional multi-element analysis techniques, pre-treatment of samples is normally not required. In most cases, the only requirement is that the sample may need to be reduced to a more suitable size for packaging and the irradiation process. Other advantages of NAA include:
Applications for NAA: Neutron activation is an established analytical technique for determining trace elements in a wide variety of materials in solid, liquid, or gaseous states. Among the potential applications for which NAA has been utilized are:
A significant advantage of NAA over other techniques is the simplicity of sample treatment before analysis: typically the only requirement is that the sample be reduced to a size suitable for encapsulation prior to conducting the irradiation procedure. Generally, a suitable sample mass may vary from milligrams to several grams of sample, depending upon the sensitivities required and specific elements to be analyzed. In order to avoid the potential for contamination, samples are weighed into vessels appropriate for the type of irradiation to be performed under Class 1000 clean room conditions. Given NAA’s excellent sensitivities, analysis is possible for nearly 70 elements, which under the appropriate experimental parameters, can also allow for the analysis of more than 30 elements on a simultaneous basis.
A significant advantage of NAA over other techniques is the simplicity of sample treatment before analysis: typically the only requirement is that the sample be reduced to a size suitable for encapsulation prior to conducting the irradiation procedure. Generally, a suitable sample mass may vary from milligrams to several grams of sample, depending upon the sensitivities required and specific elements to be analyzed. In order to avoid the potential for contamination, samples are weighed into vessels appropriate for the type of irradiation to be performed under Class 1000 clean room conditions. Given NAA’s excellent sensitivities, analysis is possible for nearly 70 elements, which under the appropriate experimental parameters, can also allow for the analysis of more than 30 elements on a simultaneous basis.
The actual irradiation process is performed by one of two methods. Samples requiring short irradiation times (< 20 min.) and low to moderate flux densities (< 1013 n cm-2s-1), are packaged in virgin polyethylene vials. Samples requiring longer irradiation times (hours or days), with moderate to high flux densities, are packaged in high-purity quartz vials. Once packaged, the samples are placed near the core of the nuclear research reactor where the neutrons emitted by the reactor interact with the nuclei of the elements’ atoms causing them to become radioactive as depicted in the diagram on the previous page.
Elemental radioisotopes are formed through this interaction that subsequently decay by emitting gamma rays (γ-rays) that are unique in half-life and energy. The distinct energy-signatures created are unique to each element and provide positive identification of the targeted element(s) in the sample, while the quantification of the element(s) are determined from the proportion of the intensity to the concentration of the respective element in the sample. Since neutrons have no charge and will pass through virtually any materials, they will interact with the nucleus of the atom, rather than the electron shell. In this respect, NAA is capable of providing a comprehensive determination of the samples total elemental content regardless of oxidation state, chemical form, or physical state of the desired element(s).
Placement or introduction of the packaged sample(s) for irradiation in the core of the research reactor is accomplished by one of two methods. For analysis of short-lived nuclides requiring shorter irradiation times (< 20 min.), polyethylene vials are placed into a larger transport device know as a rabbit. The rabbit travels from the laboratory into the reactor by an air-driven pneumatic transport system into the core of the reactor where the sample will reside until it has been subjected to the specified amount of radiation. The rabbit is then extracted from the core and transported back to the laboratory where the samples are removed from the rabbit, and transferred to non-irradiated vials for completion of the analytical process. For longer irradiation times, as many as 30 samples can be bundled into a watertight metal container called a can, which is mechanically (or manually) lowered into the reactor for the specified irradiation time. After irradiation, the can(s) are removed from the core to cool, which allows the short-lived radioactivity to dissipate to safe handling levels. The advantage of both methods is that it allows for the final analytical process to be conducted outside of the containment area, or in certain instances, by a laboratory outside the research reactor facility itself.
Upon return from the irradiation process and once the samples have been allowed to decay the specified length of time, the analyst will perform the final procedure known as counting. The counting procedure is accomplished by placement of the activated sample in a device know as a detector, which is a specialized device that looks for the resultant γ-rays emitted from the irradiation process. The detectors most commonly used for the counting procedure are either Lithium drifted Germanium [Ge(Li)] detectors, or High Purity Germanium (HPGe) detectors, with the latter being the most commonly used for multi-elemental analysis. Since γ-rays emitted from the sample are very penetrating, the resultant γ-rays will create spectra similar to that of a gas chromatograph with peaks at different retention times (as depicted in the diagram below). The position of each energy peak in the spectrograph determines the identification of the element, while the area underlying the peak is proportional to the concentration of the respective element.
Verifying the accuracy and quantification results for the NAA procedure is as important as with any viable analytical technique. While in theory, it is feasible to calculate the target atoms mathematically (or perform absolute activation analysis), it is generally not acceptable since there can be significant uncertainties in values available for various nuclear parameters. Equally important is the fact that the neutron energy spectrum and flux densities of the reactor are not always constant and can vary during different irradiation periods. For these reasons, the comparator method is frequently incorporated, whereby a standard containing a known amount of the specified element is irradiated and counted along with the subject sample(s). Since factors such as neutron flux, irradiation times, and other variables can be accounted for in the counting process, the results of the comparator standard can be used to verify the quantification and accuracy of the subject sample(s). Since the creation of individual standards can become tedious as well as cost prohibitive stoichiometrically well-defined compounds can be obtained from sources that specialize in the preparation of standard reference materials (SRMs) such as the National Institute of Standards and Technology (NIST) and the U.S. Geological Society (USGS). In this regard, either multi-elemental standards or the use of a single comparator standard for all elements can be incorporated for verification and quality control purposes.
A major advantage of NAA methods for both simple and complicated matrices is that the techniques available can be made highly selective, and that operational parameters can be exploited so that maximum sensitivity can be achieved for the desired element(s). Variation of the type, energy, or flux of the irradiating neutrons allows for the selective analysis of certain elements to be enhanced. Differences in half-lives among activated element species can also be exploited as a means to discriminate among elements in the counting process. Since a single nuclear activation technique is not applicable to every element, EAI offers clients the most comprehensive array of NAA procedures available by any other commercial laboratory. Among the types of NAA and element-specific forms available are the following procedures:
Instrumental Neutron Activation Analysis (INAA) is the most common form of activation analysis offered and utilized for individual and multi-elemental analysis. Instrumental indicates that no chemical pretreatment is performed on the sample prior to the irradiation process. The samples are simply packaged, irradiated for the specified length of time, allowed to decay, then counted, and the element results verified and reported.
Radiochemical Neutron Activation Analysis (RNAA) is another type of NAA in which the sample irradiation is followed by chemical processing or treatment. This process is designed to separate specific elements or groups of elements so that ultimate selectivity and sensitivity for the desired element is achieved. Since the chemistry is performed after the irradiation process, there is no opportunity for sample contamination as only the desired element will be radioactive.
Epithermal Neutron Activation Analysis (ENAA) is a process where neutrons having specific energy levels (from 0.1 to 1.0eV) are utilized to suppress interfering activities from product radionuclides produced in high yields from undesired elements of interest with large thermal neutron cross sections. By using Cadmium or Boron to shield the sample from these thermal neutrons (0.025eV), the sample is exposed to neutrons with higher energy values (> 0.55eV). Applications for ENAA are geological and biological samples to reduce high thermal neutron activities from elements such as Sodium, providing for lower limits of detection.
Prompt-gamma Neutron Activation Analysis (PGNAA) is based on the detection of prompt γ-rays emitted from nuclei in an excited state during irradiation rather than afterwards. Similar to other forms of NAA, the energy of the γ-ray is indicative of the isotope present and the intensity of the γ-ray is a measure of the concentration of the isotope in the sample. PGNAA is particularly useful for the determination of Hydrogen, Boron, Cadmium, or Gadolinium.
Fast Neutron Activation Analysis (FNAA) is based on reactions with high-energy (14MeV) neutrons produced by a specialized small accelerator known as a neutron generator. FNAA is a rapid non-destructive technique used for the determination of lighter elements such as Nitrogen and Oxygen, and can routinely achieve precisions of 1-2% RSD of complex matrices.

Family owned and operated since 1979 (formerly known as EAC), we strive to give each customer the best service possible.
From Aerospace to Pharmaceuticals, we will work with you to tailor your specific analytical method.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.
Please call us at 859-254-5115 or email Info@ElementalAnalysis.com if you have any questions.